What are PFAS?
Per- and Poly-FluoroAlkyl Substances (PFAS) is a very broad class of fluorinated carbon structures. There are conceptually thousands of individual PFAS, although only a relatively small percentage have been made commercially. Most of the individual PFAS types are generated as byproducts.
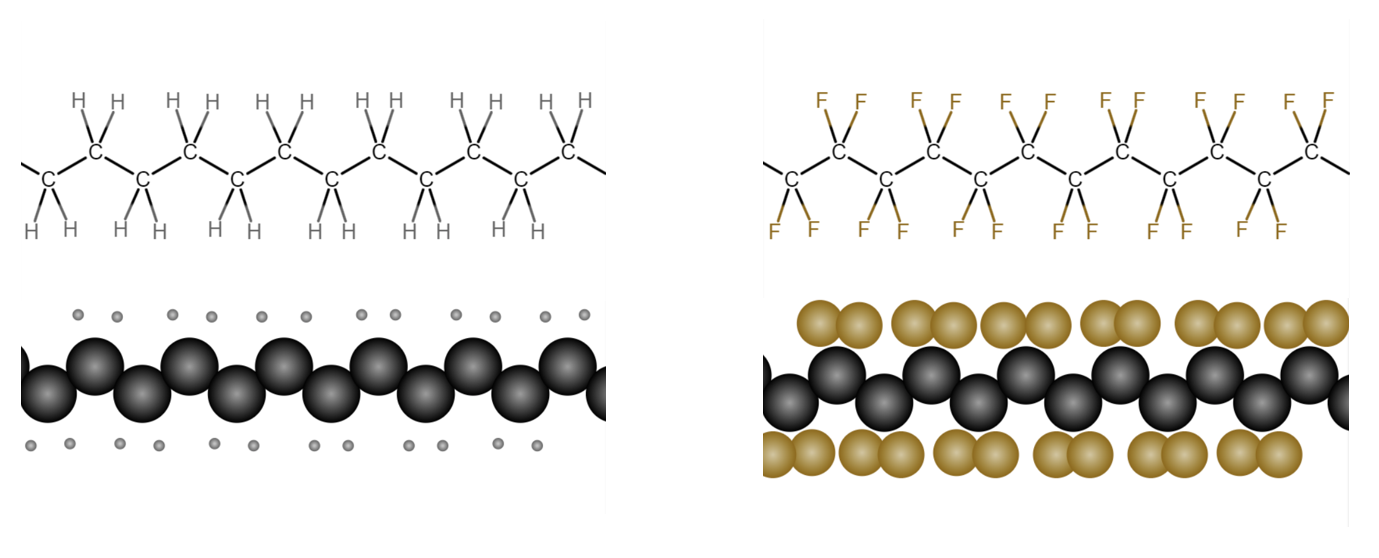
Carbon is fluorinated when a hydrogen is replaced by fluorine thru a synthetic chemical process. In other words, fluorination occurs during a laboratory or manufacturing process. There are very few naturally occurring fluorocarbons.
Poly fluorinated means that two or more hydrogen atoms bound to carbon on a molecule have been replaced by fluorine. Perfluorination means all available hydrogens on a molecule have been replaced by fluorine.
The carbon-fluorine bond is one of the strongest chemical bonds in nature, which requires a great amount of energy to break. The synthetic fluorination process applies primarily to carbon chains, i.e., many carbon atoms linked together (as opposed to carbon ring structures). The perfluorinated / polyfluorinated carbon chain is referred to as a fluorocarbon; fluorocarbons having surfactant properties are called fluorosurfactants.
Since carbon can form up to four bonds with other elements, each carbon in a chain is bound to one, two or three fluorine atoms (depending on its position in the chain and the degree of chain branching). What all this means is that a PFAS carbon chain has a densely packed fluorine exterior
that is virutally unreactive and very resistant to chemical degradation.
The unreactive nature of fluorocarbons has led to widespread, beneficial uses in many applications.
Fabric and tile surfaces are made to be stain resistant by applying a fluorocarbon layer.
Paints and printing inks form a uniform coating layer over substrates due to the wettability imparted by fluorocarbon ingredients.
Similarly, certain fire-fighting foams with fluorocarbon ingredients (e.g., AFFF) are able to rapidly cover a liquid fuel fire for fast extinguishment and prevention of re-ignition, thereby saving property and human life.
The unreactive nature of fluorocarbons also has a significant drawback. PFAS can exist in the environment for a long time, although not all PFAS types are the same. Longer chain PFAS are more bio-accumulative and toxic. Some PFAS types can degrade into other PFAS types that are of concern.
If improperly managed, the beneficial aspects of PFAS-containing materials can be overshadowed by exposure and environmental concerns. This is the subject of a vigorous and wide-ranging debate currently. Many manufacturers and suppliers choose to avoid the matter by simply providing certificates to customers that their products are PFAS-free.
Contact
What can we help you with?
-
Deliveries:
Sample Receiving
605 South 6th Ave
Mansfield, TX 76063 -
Payments:
Accounts Receivable
PO Box 1769
Mansfield, TX 76063
